Charge on an electron is given below in the table in different units. As the elementary charge is denoted by e, the electron is commonly symbolized by e. Positive and negative are just common terms there is nothing remarkable about being positively charged as opposed to negatively charged. The electrical charge on an electron is equal to the charge on proton, but with opposite sign. Answer (1 of 57): One of the basic properties of matter is electrical charge. In atom s, the electron carries a negative elementary or unit charge the proton. The electron charge to mass ratio was an experiment that was used to calculate the ratio of the electrons charge to its mass. It is used as a standard unit of charge for subatomic particles. charge (electric charge): In physics, charge, also known as electric charge, electrical charge, or electrostatic charge and symbolized q, is a characteristic of a unit of matter that expresses the extent to which it has more or fewer electrons than protons. Structure of an atom is given below – Image will be uploaded soon Charge on an ElectronĮlectric charge on an electron is -1.602 10 -19 C (or experimental value -1.602176634 10 -19 Coulombs). Electrons are negatively charged subatomic particles while proton are positive, and neutron are neutral. Protons and neutrons are found in the nucleus of the atom while electron is found in the shells or orbits around the nucleus. It is made up of three subatomic particles called electron, proton and neutron. Slowly with time, experiments and discoveries scientists proposed that atom is not the ultimate particle. charged metallic atom to next but in solution the electron is carried by the. The present accepted value of electrical charge is 1.6022 x 10 -19 C. 1-le found that the charge on the electron to be- 1.6 x 10 -19 C.

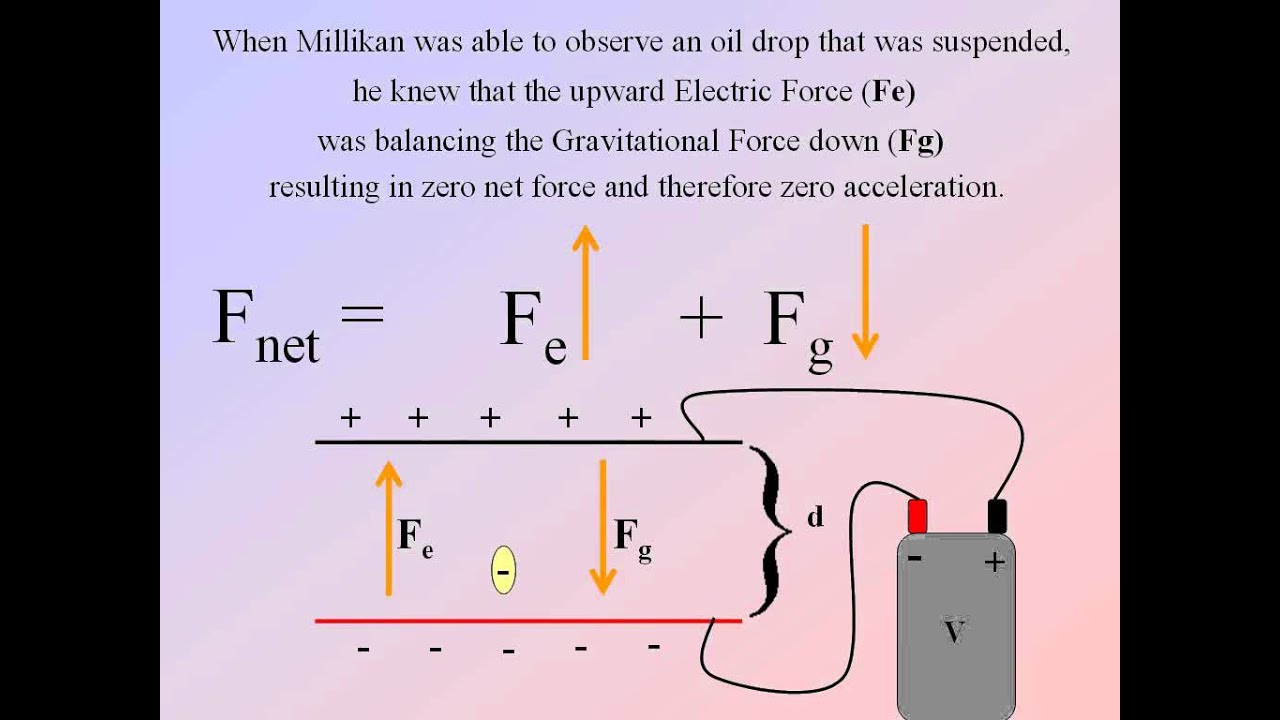
Democritus proposed that matter is made up of small units called atoms. Electric current (electricity) is a flow or movement of electrical charge. Problems of producing multiple-charged ions for electron-beam ion sources (EBIS) and for relativistic electron rings have been examined in detail in 6, 15. Millikan (1868 1953) devised a method known as oil drop experiment (1906-14), to determine the charge on the electrons.
